 (a) A sample of excited hydrogen atoms emits a characteristic red light. Each element has a unique number of electrons in a unique configuration therefore each element has its own. The electrons surrounding the atomic nucleus are arranged in a series of levels of increasing energy. You have learnt previously about the structure of an atom. Nuclear spin (I) is the total angular momentum of the nucleus of an atom. These so called line spectra are characteristic of the atomic composition of the gas. 12.3 Emission and absorption spectra (ESCQR) Emission spectra. This hyperfine structure in atomic spectra was the first evidence for nuclear spin (Herzberg, 1944). The negative sign in Equation 7.3.3 indicates that the electron-nucleus pair is more tightly bound when they are near each other than when they are far apart. For example, the 412.2 nm line of atomic Bi is actually a group of four lines falling within a width of only 0.044 nm. The orbit with n = 1 is the lowest lying and most tightly bound. In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. 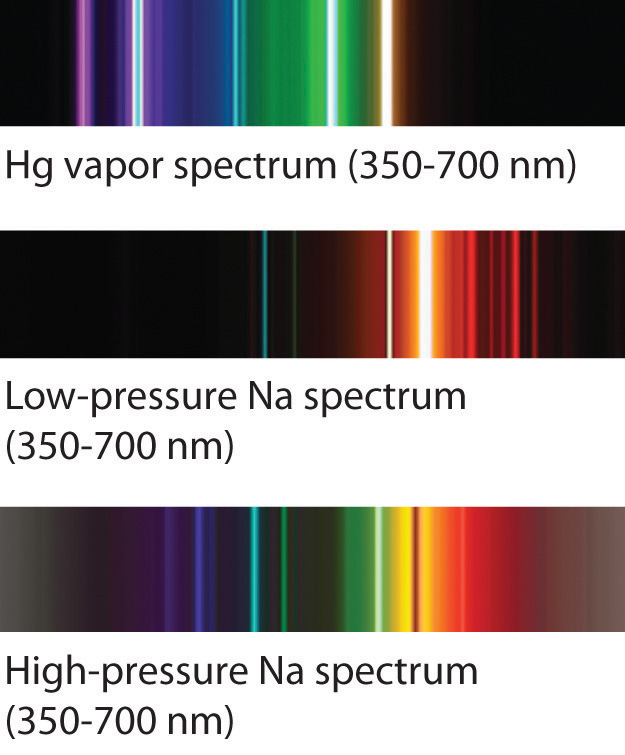
(b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. In forensic science, these techniques are of utmost importance. Atomic spectroscopy mainly consists of two types of techniques: atomic absorption spectroscopy and atomic emission spectroscopy. Hydrogen-like atoms are those atoms with only one electron remaining, regardless of the number of protons in the nucleus. The atom has been ionized.įigure 7.3.2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Atomic spectroscopy is an excellent analytical tool used for the detection and measurement of elements in a sample with high precision and confidence. 
In this state the radius of the orbit is also infinite. In that level, the electron is unbound from the nucleus and the atom has been separated into a negatively charged (the electron) and a positively charged (the nucleus) ion. Line emission spectra (atomic spectra) can be used to identify the presence of an element in a sample because no two elements produce the same line emission (atomic) spectrum. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. The line emission (atomic) spectrum of an element is unique. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
